Fetal and transitional circulation
Fetal and transitional circulation
Fetal circulation is different from adult circulation. The changes occurring soon after birth constitutes transitional circulation. Respiratory gas exchange in the fetus occurs in the placenta rather than the lungs. Fetal cardiovascular system is designed so that the most saturated blood reaches the heart and the brain. Fetal circulation can be called a shunt dependent circulation because there are intracardiac and extracardiac shunts [1].
Cardiac output in the fetus is called combined ventricular output (CVO). Despite the low oxygen partial pressures in fetal blood, presence of fetal hemoglobin and high combined ventricular output helps in the oxygen delivery to the tissues. Fetal blood returns to the placenta by the umbilical arteries and returns to the fetus by the umbilical vein. More than half of this bypasses the hepatic circulation through the ductus venosus and reaches the inferior vena cava.
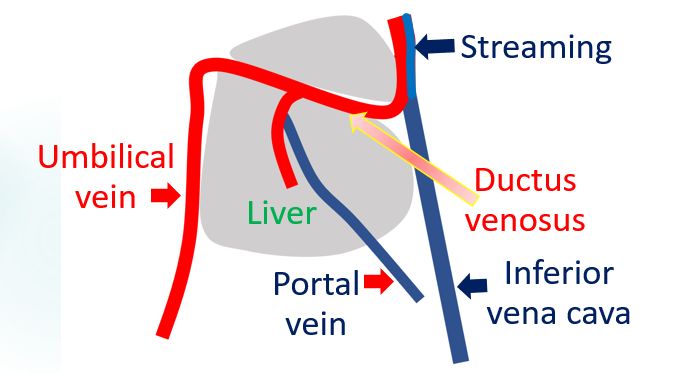
Streaming of blood from the ductus venosus with saturation of 80-90% is separate from the desaturated blood returning from the lower part of the body. Eustachian valve at the junction of the inferior vena cava and right atrium directs this saturated blood to the foramen ovale and into the left atrium.
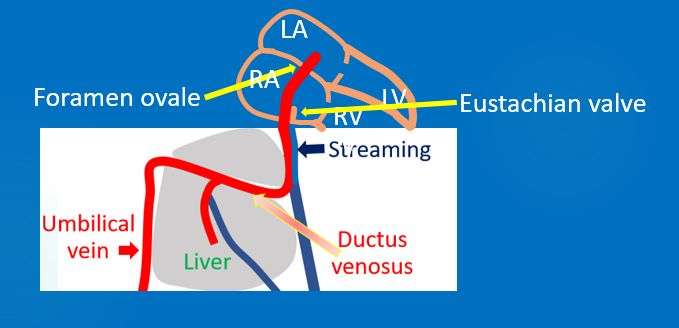
Left atrial oxygen saturation is about 65% in the fetus. This saturated blood passes into the left ventricle and is ejected into the aorta. Major part of this blood reaches the cerebral and coronary circulation. Desaturated blood from the superior vena cava, coronary sinus and the remaining part of the inferior vena caval stream is directed across the tricuspid valve into the right ventricle. Only around 12% of the blood ejected from the right ventricle reaches the lung due to high fetal pulmonary vascular resistance. Rest of it crosses the ductus arteriosus and reaches descending aorta. So lower body gets relatively desaturated blood. Part of it goes to the placenta for oxygenation.
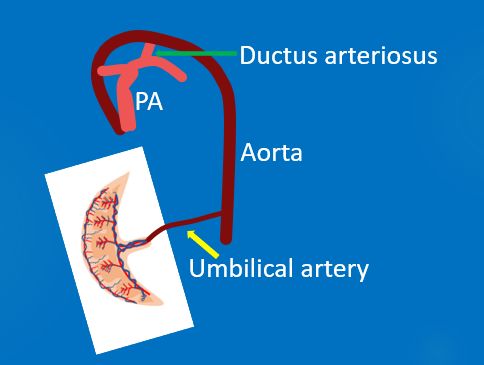
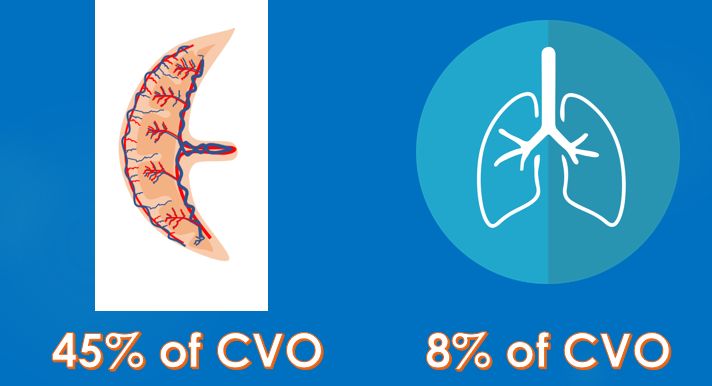
In the fetus, 65% of the venous return reaches the right ventricle and 35% reaches the left ventricle, due to the shunts. About 45% of the combined ventricular output is delivered to the placenta while the lung receives only 8%.
Soon after the birth lung expands and lung liquid is cleared. This leads to a marked fall in pulmonary vascular resistance and increase in pulmonary blood flow [2]. The fetal right ventricular dominance changes to left ventricular dominance over a period of time as the right ventricular muscle mass regresses and left ventricular muscle mass increases to support the systemic vascular resistance.
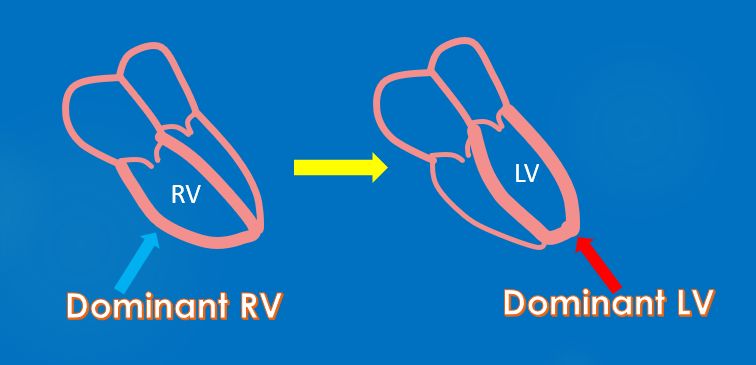
Increase in pulmonary venous return raises left atrial pressure and the valvular foramen ovale closes. Ductus arteriosus starts constricting on exposure to the high oxygen saturation in blood after birth. Ductus venosus also gets obliterated.
Transitional circulation is relevant in congenital heart diseases. The closure of ductus arteriosus in the early neonatal period can lead to marked fall in the oxygen saturation in cyanotic congenital heart disease with low pulmonary blood flow. Maintaining ductal patency with prostaglandin infusion or ductal stenting is often resorted to as a bail out procedure in such situations. There is a similar role for maintaining ductal patency in ductus dependent systemic circulation as well.
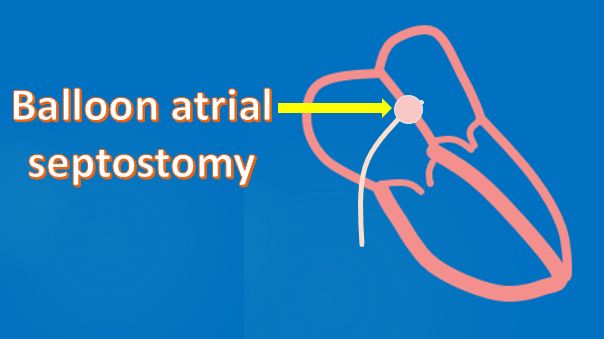
Similarly, a restrictive foramen ovale leads to desaturation in transposition of great arteries. Here comes the role of neo-natal balloon atrial septostomy in dextro transposition of great arteries.
In case of atrial septal defect, significant left to right shunt is usually not established soon after birth. Maximum shunting occurs when the right ventricular muscle mass regresses and right ventricular compliance increases. This was why the clinical diagnosis of atrial septal defect is often delayed beyond early infancy. Even with ventricular septal defect and patent ductus arteriosus, the full establishment of the left to right shunt is delayed a few weeks, awaiting the regression of pulmonary vascular resistance. That is why these lesions may not be picked up clinically soon after birth and needs follow up evaluation so that they are not missed.
Closure of both ductus arteriosus and ductus venosus is delayed in preterm infants [3]. Prevalence of patent ductus arteriosus increases with increasing prematurity. It is said that the premature ductus responds poorly to oxygen and more to prostaglandin. Clamping of the umbilical cord after delivery leads a 30-50% decrease in venous return [4]
References
- John PJ. The fetal circulation. Contin Educ Anaesth Crit Care Pain. 2005;5(4):107:112.
- Rudolph AM. The changes in the circulation after birth. Their importance in congenital heart disease. Circulation. 1970 Feb;41(2):343-59.
- Kondo M, Itoh S, Kunikata T, Kusaka T, Ozaki T, Isobe K, Onishi S. Time of closure of ductus venosus in term and preterm neonates. Arch Dis Child Fetal Neonatal Ed. 2001 Jul;85(1):F57-9.
- van Vonderen JJ, Roest AA, Siew ML, Walther FJ, Hooper SB, te Pas AB. Measuring physiological changes during the transition to life after birth. Neonatology. 2014;105(3):230-42.
