Risk stratification in cardiac channelopathies
Risk stratification in cardiac channelopathies
Risk stratification in cardiac channelopathies:
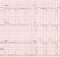
LQTS
In children, there is no particular difference between LQT types regarding the risk of SCD. The risk is more for type 2 after the age of 18 years, while the higher risk in type 3 is after 40 years. Exercise testing has no significant role in risk stratification.
Aborted SCD is a very important risk factor. QTc > 500 confers higher risk and so does a cardiac event before the age of 12 months. Family history of SCD was not important regardless of genotype. Males are at higher risk in the age group below 10 years while above that females are at a higher risk. Transmembrane mutations confer a higher risk in LQT1. If more than 90% loss of function, the risk is definitely higher. Higher risk when associated sensorineural deafness or other somatic defects as in Timothy syndrome. Certain single gene polymorphisms are also associated with higher risk.
In childhood genotyping does not give an added advantage in predicting the outcome as the phenotype is more important. Majority of the gene carriers are asymptomatic. Majority of treatment failures are due to non compliance. Only advantage of genotyping is that treatment should be started before 5 years in LQT1, later in LQT2 and still later in LQT3.
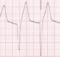
Brugada syndrome
Type I ECG is an important factor in conveying high risk. General consensus is that EP studies do not predict SCD. Conventional stimulation protocols may not be enough and epicardial studies may be required as the abnormality is more in the right outflow tract region.
No male preponderance was noted in children in the symptomatic group. In paediatric group, SVT is as common as VT. Fever is known to precipitate events in Brugada syndrome. Quinidine seems to effective in small series in paediatric age group, at least as bridge to ICD.
CPVT
Beta blockers alone is not effective in 30% of cases. ICDs may be poorly tolerated because of inappropriate shocks.