Tetralogy of Fallot (TOF)
Tetralogy of Fallot (TOF)
The four cardinal features of tetralogy of Fallot (TOF) are:
- Malalignment ventricular septal defect (VSD)
- Overriding aorta
- Infundibular pulmonary stenosis
- Right ventricular hypertrophy
The variability in clinical presentation of TOF correlates with degree of right ventricular outflow tract (RVOT) obstruction and the size/anatomy of pulmonary artery and its branches.
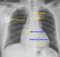
Tetralogy of Fallot – Right aortic arch
The lifted up apex (cor en sabot – peasant’s boot shaped heart) due to the right ventricular hypertrophy is seen well in this chest X-ray PA view. The right sided aortic arch is seen intending the tracheal air column on the right side. There is mild cardiomegaly and right atrial enlargement as well in this adult person with tetralogy of Fallot and associated inferior wall myocardial infarction. The lung fields are oligemic due to the right ventricular outflow tract obstruction in Tetralogy of Fallot. Tetralogy of Fallot is the commonest cause of right aortic arch in an adult.
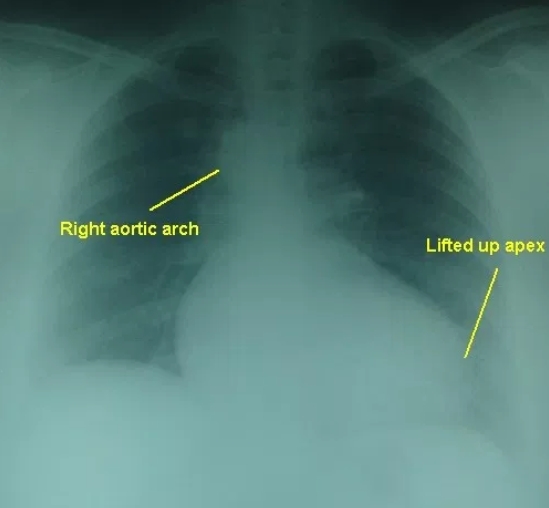
Colour Doppler echocardiography in Tetralogy of Fallot in parasternal long axis view
The blue colour is the flow of blood from right ventricle (RV) across the ventricular septal defect into the overriding aorta. This causes desaturation of aortic blood and cyanosis in Tetralogy of Fallot. The blood from the right ventricle preferentially enters the aorta which is overriding the ventricular septal defect (VSD) because the right ventricular outflow tract is narrowed in Tetralogy of Fallot as result of infundibular stenosis. The live colour Doppler video below shows the blue flow from RV to aorta across the VSD. Encoding in colour Doppler is blue for flow away from the transducer (located at the top of the sector) and red for flow towards the transducer.
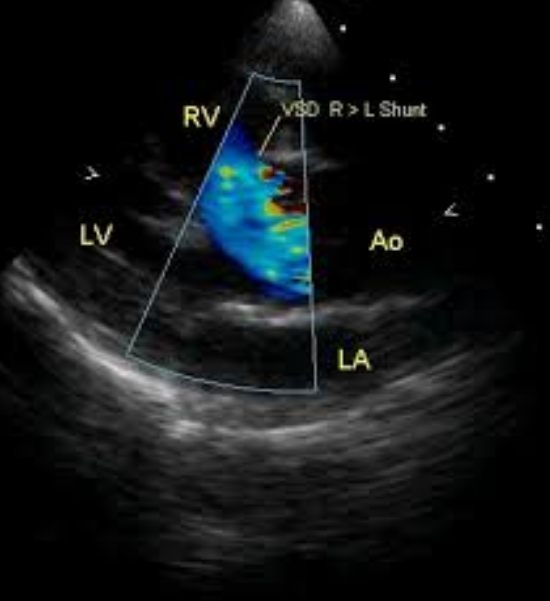
Parasternal long axis view in Tetralogy of Fallot, in diastole
Parasternal long axis view in Tetralogy of Fallot, in diastole
Parasternal long axis (PLAX) view in Tetralogy of Fallot, diastolic frame showing the aortic valve in closed position and mitral valve in open position. The aortic valve appears to impinge on the ventricular septum, but the ventricular septal defect (VSD) with aortic override and connection between right ventricle (RV) and aorta (Ao) is evident just above the septum. LA: left atrium; LV: left ventricle; IVS: interventricular septum; AML: anterior mitral leaflet.
Apical five chamber view in Tetralogy of Fallot
Apical five chamber view in Tetralogy of Fallot demonstrating the subaortic ventricular septal defect (VSD) with aortic override. 50% of the aorta (Ao) is committed to the left ventricle (LV) while the remaining half is committed to the right ventricle (RV). LA: left atrium; RA: right atrium. The VSD in Tetralogy of Fallot is a malalignment VSD which results from the malalignment of the ventricular septum with respect to the aorticopulmonary septum during embryonic development. The shift of the aorticopulmonary septum towards the pulmonary side produces both the ventricular septal defect and the narrowing of the right ventricular outflow tract.
Van Praagh R et al [1] hypothesized that all the features of tetralogy of Fallot are due to the underdevelopment of the infundibular septum and its sequelae. They termed this as a ‘monology‘, called by others as Van Praagh’s monology of Fallot!
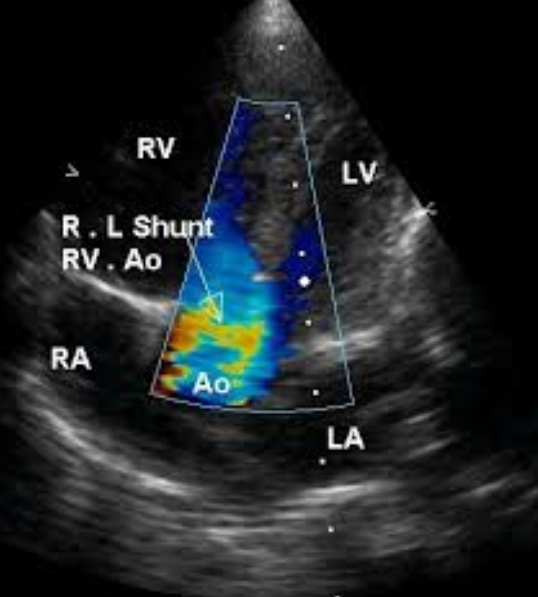
Apical five chamber view in Tetralogy of Fallot with colour flow mapping (Colour Doppler imaging) in systole with right to left shunt across the VSD. Blue stream moving from right ventricle across the VSD to the aorta is clearly visualised in this frame. There is also a blue stream from the left ventricle to the aorta.
Patent ductus arteriosus jet in Tetralogy of Fallot on Colour Doppler imaging
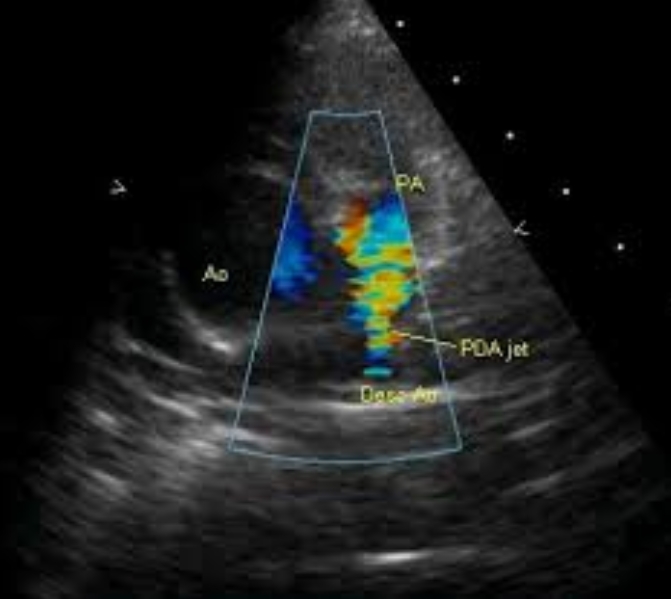
Colour flow imaging shows high velocity jet in the pulmonary artery arising distally, from the descending aorta, suggesting a patent ductus arteriosus (PDA). This is one of the compensatory mechanisms to improve pulmonary flow in Tetralogy of Fallot. Another mechanism is major aortopulmonary collateral arteries (MAPCA). Intra pulmonary collaterals can also occur in Tetralogy of Fallot. Desc Ao: descending aorta. The image is in the parasternal short axis view.
PDA Jet in Tetralogy of Fallot by Continuous wave Doppler
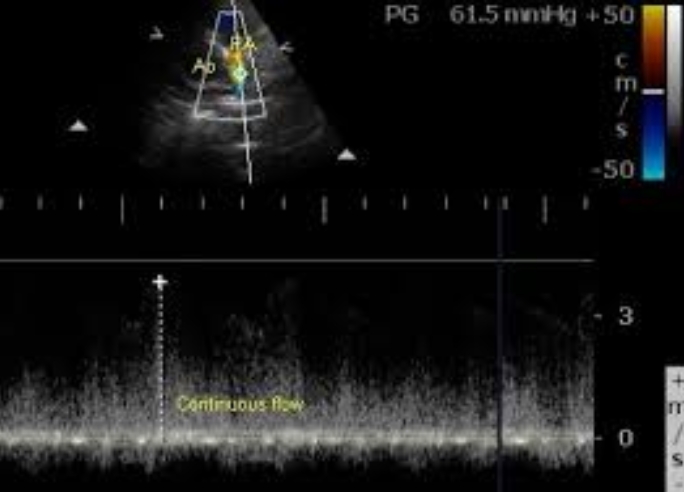
Continuous wave Doppler interrogation of the jet guided by colour flow mapping picks up the continuous flow with a peak gradient of 61.5 mm Hg. The gradient is calculated from the velocity measured by the device using the formula: P = 4 V2. Ao: aorta; PA: pulmonary artery
Findings to be sought in an aortogram in Tetralogy of Fallot
- Aortic regurgitation
- Coronary anomalies
- MAPCAS (major aortopulmonary collaterals)
- PDA (patent ductus arteriosus)
- Side of the aortic arch
MAPCA from right internal mammary artery (RIMA)
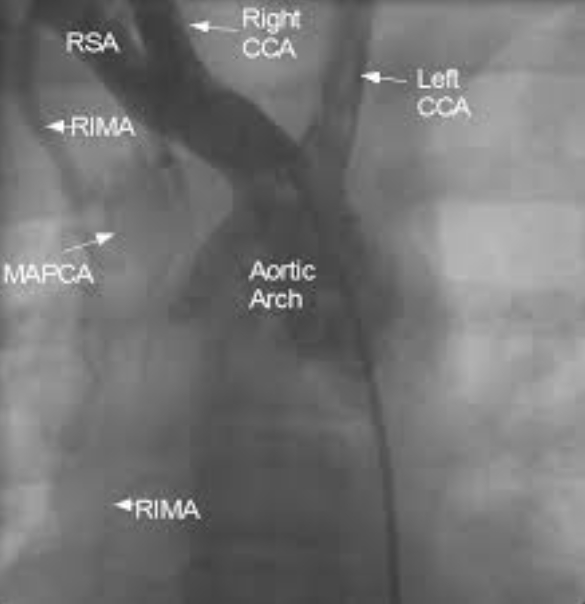
Still frame from an angiogram with radiocontrast dye injected using a pigtail catheter kept in the right brachiocephalic artery showing major aortopulmonary collateral artery (MAPCA) arising from the right internal mammary artery (RIMA). RSA: right subclavian artery; Right CCA: right common carotid artery; Left CCA: left common carotid artery. Left subclavian artery is not visualised well as the dye reflux into the arch of aorta is not enough to opacify it and the proximal holes of the pigtail are beyond its origin. The second frame (below) gives a better picture of the tortuous branches of the MAPCA. MAPCAs are seen in severe forms of Tetralogy of Fallot and pulmonary atresia. MAPCAs usually arise from the descending aorta. Strictly speaking the collateral arising from RIMA is not a major “aorto” pulmonary collateral, though it can be considered a MAPCA in the wider sense of the meaning. When the lungs are supplied by multiple MAPCAs, they are unifocalised prior to definitive surgical repair of Tetralogy of Fallot. Connecting the distal end of MAPCAs to a single vessel is known as unifocalization. Collaterals to the pulmonary arterial branches can also arise from the bronchial arteries within the lungs. Hilar collaterals can also occur in pulmonary atresia.
Treatment options and sequelae
Surgical approaches to TOF would include palliative systemic – pulmonary shunts like Blalock-Taussig shunt, Waterston shunt and Potts shunt. Complete repair is accomplished by patch VSD closure, resection of subpulmonic obstruction, a transannular patch around the pulmonary valve annulus if necessary and take down of prior shunt. Placement of a transannular patch for widening of the RVOT usually leads to severe pulmonary regurgitation.
Systemic-pulmonary shunt leads to high flow through pulmonary artery, elevated pulmonary vascular resistance and branch pulmonary artery distortion. Survival after repair is worse in patients with prior central shunts (Waterston or Potts) possibly due to the higher unrestricted pulmonary blood flow. Some patients with Blalock-Taussig shunts may survive unrepaired into adulthood. These patients should be evaluated for pulmonary artery stenosis and pulmonary hypertension.
Those who had pulmonary valve atresia or anomalous left anterior descending coronary artery may have had prosthetic or homograft conduits with or without a valve placed between the right ventricle and pulmonary artery. Endothelial overgrowth can occur within the conduits and cause obstruction of the right ventricular outflow tract. This can be treated with balloon dilatation or surgical replacement of the conduit.
Mechanisms of aortic regurgitation in operated tetralogy of Fallot
Pulmonary regurgitation is almost universal after corrective repair of tetralogy of Fallot, more so in those who require a transannular patch for widening of the right ventricular outflow tract. Hence an early diastolic murmur along the left sternal edge following repair of tetralogy of Fallot is most often due to pulmonary regurgitation. But a few cases may also develop aortic regurgitation due to various reasons. Aorta is dilated in tetralogy of Fallot prior to repair because it receives a major portion of the output from the right ventricle as well as the left ventricular output. This is the reason for a high volume pulse in tetralogy of Fallot. Thus dilatation of the aortic root is one of the potential reasons for aortic regurgitation in tetralogy of Fallot. Other causes are lack of support due to a subaortic ventricular septal defect and valvular deformation resulting from retraction of the surgical patch.
Potential for sudden cardiac death after surgical repair of tetralogy of Fallot
The risk of sudden cardiac death in operated tetralogy of Fallot is 25-100 fold than in the general population and it can occur decades after correction. The risk is related to QRS duration more than 180 milliseconds. The QRS widening is related to pulmonary regurgitation, right ventricular dilatation and conduction defect. Atrial arrhythmias also common after TOF repair. Hemodynamic effects of pulmonary regurgitation include chronic right ventricular volume overload, right ventricular dysfunction and exercise intolerance. Pulmonary valve replacement can decrease QRS duration and stabilize right ventricular function, though the timing is unclear; but earlier would be better than later. Right ventricular function can be evaluated by echo or magnetic resonance imaging (MRI).
It is well known that adults with previously operated tetralogy of Fallot can develop ventricular tachyarrhythmias and die suddenly. They are prone for ventricular tachycardia as well as atrial tachyarrhythmias like atrial flutter and fibrillation. Syncope may be a forerunner of sudden death in some individuals with operated tetralogy of Fallot and calls for evaluation. An annual incidence of 0.4 percent sudden death during the first twenty five years after surgery has been reported. Both the surgical scar as well as the dilatation of right ventricle and right atrium due to the pulmonary and tricuspid regurgitation are thought to have roles in arrhythmogenesis. Highest risk is in those with marked cardiomegaly (cardiothoracic ratio more than 60 percent), severe pulmonary and or tricuspid regurgitation, QRS duration on the electrocardiogram of more than 180 milliseconds, and a QT interval dispersion of more than 60 milliseconds.
[1,2]
Surgical correction of pulmonary regurgitation with a valvular prosthesis and tricuspid regurgitation by annuloplasty may decrease the chance of atrial and ventricular arrhythmias. This is more likely if surgical repair is also accompanied by mapping and ablation of the reentry circuit of arrhythmia [3]
References
- Van Praagh R, Van Praagh S, Nebesar RA, Muster AJ, Sinha SN, Paul MH. Tetralogy of Fallot: underdevelopment of the pulmonary infundibulum and its sequelae. Am J Cardiol. 1970 Jul;26(1):25-33.
- Gatzoulis MA, Till JA, Somerville J, Redington AN. Mechanoelectrical interaction in tetralogy of Fallot: QRS prolongation relates to right ventricular size and predicts malignant ventricular arrhythmias and sudden death. Circulation 1995;92:231-7.
- Gatzoulis MA, Till JA, Redington AN. Depolarization-repolarization inhomogeneity after repair of tetralogy of Fallot: the substrate for malignant ventricular tachycardia? Circulation 1997;95:401-4.
- Therrien J, Siu SC, Harris L, Dore A, Niwa K, Janousek J, Williams WG, Webb G, Gatzoulis MA. Impact of pulmonary valve. replacement on arrhythmia propensity late after repair of tetralogy of Fallot. Circulation 2001;103: 2489-94